| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-09-30 22:41:59 UTC |
|---|
| Update Date | 2020-05-21 16:28:43 UTC |
|---|
| BMDB ID | BMDB0001170 |
|---|
| Secondary Accession Numbers | |
|---|
| Metabolite Identification |
|---|
| Common Name | Lathosterol |
|---|
| Description | Lathosterol, also known as gamma-cholesterol, belongs to the class of organic compounds known as cholesterols and derivatives. Cholesterols and derivatives are compounds containing a 3-hydroxylated cholestane core. Thus, lathosterol is considered to be a sterol. Based on a literature review a significant number of articles have been published on Lathosterol. |
|---|
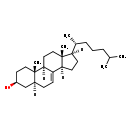
| Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| gamma-Cholesterol | ChEBI | | 5alpha-Cholest-7-en-3beta-ol | Kegg | | g-Cholesterol | Generator | | Γ-cholesterol | Generator | | 5a-Cholest-7-en-3b-ol | Generator | | 5Α-cholest-7-en-3β-ol | Generator | | (3beta)-Cholest-7-en-3-ol | HMDB | | (3beta,5alpha)-Cholest-7-en-3-ol | HMDB | | (3beta,alpha)-Cholest-7-en-3-ol | HMDB | | (7)-Cholestenol | HMDB | | 3b-Hydroxy-5-cholestene | HMDB | | 3beta-Hydroxy-5alpha-cholest-7-ene | HMDB | | 5-a-Cholest-7-en-3-beta-ol | HMDB | | 5-alpha-Cholest-7-en-3-beta-ol | HMDB | | 7-Cholesten-3-beta-ol | HMDB | | 7-Cholestenol | HMDB | | 7-Cholesterol | HMDB | | Cholest-7-en-3-ol | HMDB | | Cholest-7-en-3beta-ol | HMDB | | Cholest-7-en-ol | HMDB | | Cholesterol - synthetic | HMDB | | Cholesterol extra pure | HMDB | | Cholesterol GR | HMDB | | Cholesterol pharma | HMDB | | Cholesterol,NF grade | HMDB | | delta(7)-Cholestenol | HMDB | | DELTA7-Cholesten-3beta-ol | HMDB | | DELTA7-Cholestenol | HMDB | | gamma-Cholestenol | HMDB | | Lathosterol | ChEBI |
|
|---|
| Chemical Formula | C27H46O |
|---|
| Average Molecular Weight | 386.6535 |
|---|
| Monoisotopic Molecular Weight | 386.354866094 |
|---|
| IUPAC Name | (1R,2S,5S,7S,11R,14R,15R)-2,15-dimethyl-14-[(2R)-6-methylheptan-2-yl]tetracyclo[8.7.0.0^{2,7}.0^{11,15}]heptadec-9-en-5-ol |
|---|
| Traditional Name | (1R,2S,5S,7S,11R,14R,15R)-2,15-dimethyl-14-[(2R)-6-methylheptan-2-yl]tetracyclo[8.7.0.0^{2,7}.0^{11,15}]heptadec-9-en-5-ol |
|---|
| CAS Registry Number | 80-99-9 |
|---|
| SMILES | [H][C@@]1(CC[C@@]2([H])C3=CC[C@@]4([H])C[C@@H](O)CC[C@]4(C)[C@@]3([H])CC[C@]12C)[C@H](C)CCCC(C)C |
|---|
| InChI Identifier | InChI=1S/C27H46O/c1-18(2)7-6-8-19(3)23-11-12-24-22-10-9-20-17-21(28)13-15-26(20,4)25(22)14-16-27(23,24)5/h10,18-21,23-25,28H,6-9,11-17H2,1-5H3/t19-,20+,21+,23-,24+,25+,26+,27-/m1/s1 |
|---|
| InChI Key | IZVFFXVYBHFIHY-SKCNUYALSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as cholesterols and derivatives. Cholesterols and derivatives are compounds containing a 3-hydroxylated cholestane core. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Lipids and lipid-like molecules |
|---|
| Class | Steroids and steroid derivatives |
|---|
| Sub Class | Cholestane steroids |
|---|
| Direct Parent | Cholesterols and derivatives |
|---|
| Alternative Parents | |
|---|
| Substituents | - Cholesterol-skeleton
- Cholesterol
- 3-beta-hydroxysteroid
- Hydroxysteroid
- 3-hydroxysteroid
- 3-hydroxy-delta-7-steroid
- Delta-7-steroid
- Cyclic alcohol
- Secondary alcohol
- Organic oxygen compound
- Hydrocarbon derivative
- Organooxygen compound
- Alcohol
- Aliphatic homopolycyclic compound
|
|---|
| Molecular Framework | Aliphatic homopolycyclic compounds |
|---|
| External Descriptors | |
|---|
| Ontology |
|---|
| Status | Expected but not Quantified |
|---|
| Origin | |
|---|
| Biofunction | Not Available |
|---|
| Application | Not Available |
|---|
| Cellular locations | - Cell membrane
- Cytoplasm
- Endoplasmic reticulum
- Membrane
- Myelin sheath
|
|---|
| Physical Properties |
|---|
| State | Solid |
|---|
| Experimental Properties | | Property | Value | Reference |
|---|
| Melting Point | 122.0 °C | Not Available | | Boiling Point | Not Available | Not Available | | Water Solubility | Not Available | Not Available | | LogP | Not Available | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-0ab9-1019000000-45e8f26aff61be3d325a | View in MoNA |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (1 TMS) - 70eV, Positive | splash10-0006-3004900000-efe936a9ed1a83aa429c | View in MoNA |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | View in JSpectraViewer |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-014r-0009000000-88fe25051d65e99ecb32 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-05p9-3149000000-d25373f7179ed89b61dc | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0c00-6359000000-8bff83f3efe22597deab | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-000i-0009000000-82b1527837d67c6be40a | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-000i-0009000000-e2badd995ee41a69b18f | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0ldi-1009000000-96f6e67651380e1c959e | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-000i-0009000000-178681247f5078239898 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0avu-9233000000-247b9ee1da8d484a4da6 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0a4l-9710000000-8ebbaf147cf1232af879 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-000i-0009000000-c037f2d2f217e99b79ac | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-000i-0009000000-c037f2d2f217e99b79ac | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-001i-0009000000-fbd8e8ecfb898bb732a5 | View in MoNA |
|---|
| 1D NMR | 1H NMR Spectrum (1D, 600 MHz, CDCl3, experimental) | Not Available | View in JSpectraViewer |
|---|
| 2D NMR | [1H, 13C]-HSQC NMR Spectrum (2D, 600 MHz, CDCl3, experimental) | Not Available | View in JSpectraViewer |
|---|
|
|---|