| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-09-30 23:21:47 UTC |
|---|
| Update Date | 2020-04-22 15:17:22 UTC |
|---|
| BMDB ID | BMDB0006354 |
|---|
| Secondary Accession Numbers | |
|---|
| Metabolite Identification |
|---|
| Common Name | Deoxythymidine diphosphate-l-rhamnose |
|---|
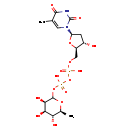
| Description | Deoxythymidine diphosphate-l-rhamnose, also known as thymidine diphosphoric acid rhamnose or dTDP-L-rhamnose, belongs to the class of organic compounds known as pyrimidine nucleotide sugars. These are pyrimidine nucleotides bound to a saccharide derivative through the terminal phosphate group. Deoxythymidine diphosphate-l-rhamnose is an extremely weak basic (essentially neutral) compound (based on its pKa). Deoxythymidine diphosphate-l-rhamnose exists in all living organisms, ranging from bacteria to humans. |
|---|
| Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| Thymidine 5'-(trihydrogen diphosphate), p'-(6-deoxy-beta-L-mannopyranosyl) ester | ChEBI | | Thymidine diphosphate rhamnose | ChEBI | | Thymidine diphosphate-L-rhamnose | ChEBI | | Thymidine 5'-(trihydrogen diphosphate), p'-(6-deoxy-b-L-mannopyranosyl) ester | Generator | | Thymidine 5'-(trihydrogen diphosphate), p'-(6-deoxy-β-L-mannopyranosyl) ester | Generator | | Thymidine 5'-(trihydrogen diphosphoric acid), p'-(6-deoxy-b-L-mannopyranosyl) ester | Generator | | Thymidine 5'-(trihydrogen diphosphoric acid), p'-(6-deoxy-beta-L-mannopyranosyl) ester | Generator | | Thymidine 5'-(trihydrogen diphosphoric acid), p'-(6-deoxy-β-L-mannopyranosyl) ester | Generator | | Thymidine diphosphoric acid rhamnose | Generator | | Thymidine diphosphoric acid-L-rhamnose | Generator | | Deoxythymidine diphosphoric acid-L-rhamnose | Generator | | dTDP-6-Deoxy-L-mannose | HMDB | | dTDP-L-Rhamnose | HMDB |
|
|---|
| Chemical Formula | C16H26N2O15P2 |
|---|
| Average Molecular Weight | 548.3296 |
|---|
| Monoisotopic Molecular Weight | 548.080841196 |
|---|
| IUPAC Name | {[hydroxy({[(2R,3S,5R)-3-hydroxy-5-(5-methyl-2,4-dioxo-1,2,3,4-tetrahydropyrimidin-1-yl)oxolan-2-yl]methoxy})phosphoryl]oxy}({[(3R,4R,5R,6S)-3,4,5-trihydroxy-6-methyloxan-2-yl]oxy})phosphinic acid |
|---|
| Traditional Name | dtdp-L-rhamnose |
|---|
| CAS Registry Number | 2147-59-3 |
|---|
| SMILES | C[C@@H]1OC(OP(O)(=O)OP(O)(=O)OC[C@H]2O[C@H](C[C@@H]2O)N2C=C(C)C(=O)NC2=O)[C@H](O)[C@H](O)[C@H]1O |
|---|
| InChI Identifier | InChI=1S/C16H26N2O15P2/c1-6-4-18(16(24)17-14(6)23)10-3-8(19)9(31-10)5-29-34(25,26)33-35(27,28)32-15-13(22)12(21)11(20)7(2)30-15/h4,7-13,15,19-22H,3,5H2,1-2H3,(H,25,26)(H,27,28)(H,17,23,24)/t7-,8-,9+,10+,11-,12+,13+,15?/m0/s1 |
|---|
| InChI Key | ZOSQFDVXNQFKBY-CZRCVJRHSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as pyrimidine nucleotide sugars. These are pyrimidine nucleotides bound to a saccharide derivative through the terminal phosphate group. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Nucleosides, nucleotides, and analogues |
|---|
| Class | Pyrimidine nucleotides |
|---|
| Sub Class | Pyrimidine nucleotide sugars |
|---|
| Direct Parent | Pyrimidine nucleotide sugars |
|---|
| Alternative Parents | |
|---|
| Substituents | - Pyrimidine nucleotide sugar
- Pyrimidine 2'-deoxyribonucleoside diphosphate
- Pentose phosphate
- Monosaccharide phosphate
- Organic pyrophosphate
- Monoalkyl phosphate
- Pyrimidone
- Hydropyrimidine
- Phosphoric acid ester
- Monosaccharide
- Organic phosphoric acid derivative
- Oxane
- Alkyl phosphate
- Pyrimidine
- Heteroaromatic compound
- Vinylogous amide
- Tetrahydrofuran
- Urea
- Lactam
- Secondary alcohol
- Oxacycle
- Azacycle
- Organoheterocyclic compound
- Polyol
- Organonitrogen compound
- Alcohol
- Hydrocarbon derivative
- Organic oxide
- Organopnictogen compound
- Organooxygen compound
- Organic nitrogen compound
- Organic oxygen compound
- Aromatic heteromonocyclic compound
|
|---|
| Molecular Framework | Aromatic heteromonocyclic compounds |
|---|
| External Descriptors | |
|---|
| Ontology |
|---|
| Status | Expected but not Quantified |
|---|
| Origin | |
|---|
| Biofunction | Not Available |
|---|
| Application | Not Available |
|---|
| Cellular locations | |
|---|
| Physical Properties |
|---|
| State | Solid |
|---|
| Experimental Properties | | Property | Value | Reference |
|---|
| Melting Point | Not Available | Not Available | | Boiling Point | Not Available | Not Available | | Water Solubility | Not Available | Not Available | | LogP | Not Available | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-0036-6922340000-5b7bbefa46231e0401e7 | View in MoNA |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (2 TMS) - 70eV, Positive | splash10-00bc-8441509000-f72396e274d70b76dce1 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-004i-0900000000-a235a91a2d2afe7e5b0e | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-004i-3921000000-f27277a5a22d8b1a6e62 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-004i-4900000000-385a0d128de7afa0ef56 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0f6t-3303290000-8fb8bdb50a4d4dd37d52 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0ufu-5915010000-0710c988c191bfa8f10b | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0a6u-4901000000-ac82c124189fc9d5bf1d | View in MoNA |
|---|
| 1D NMR | 13C NMR Spectrum (1D, 100 MHz, H2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 1H NMR Spectrum (1D, 100 MHz, H2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 13C NMR Spectrum (1D, 1000 MHz, H2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 1H NMR Spectrum (1D, 1000 MHz, H2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 13C NMR Spectrum (1D, 200 MHz, H2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 1H NMR Spectrum (1D, 200 MHz, H2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 13C NMR Spectrum (1D, 300 MHz, H2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 1H NMR Spectrum (1D, 300 MHz, H2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 13C NMR Spectrum (1D, 400 MHz, H2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 1H NMR Spectrum (1D, 400 MHz, H2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 13C NMR Spectrum (1D, 500 MHz, H2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 1H NMR Spectrum (1D, 500 MHz, H2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 13C NMR Spectrum (1D, 600 MHz, H2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 1H NMR Spectrum (1D, 600 MHz, H2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 13C NMR Spectrum (1D, 700 MHz, H2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 1H NMR Spectrum (1D, 700 MHz, H2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 13C NMR Spectrum (1D, 800 MHz, H2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 1H NMR Spectrum (1D, 800 MHz, H2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 13C NMR Spectrum (1D, 900 MHz, H2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 1H NMR Spectrum (1D, 900 MHz, H2O, predicted) | Not Available | View in JSpectraViewer |
|---|
|
|---|