| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-09-30 23:48:30 UTC |
|---|
| Update Date | 2020-06-04 19:40:02 UTC |
|---|
| BMDB ID | BMDB0007988 |
|---|
| Secondary Accession Numbers | |
|---|
| Metabolite Identification |
|---|
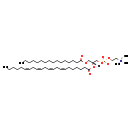
| Common Name | PC(16:0/22:4(7Z,10Z,13Z,16Z)) |
|---|
| Description | PC(16:0/22:4(7Z,10Z,13Z,16Z)), also known as phosphatidylcholine(16:0/22:4) or gpcho(16:0/22:4), belongs to the class of organic compounds known as phosphatidylcholines. These are glycerophosphocholines in which the two free -OH are attached to one fatty acid each through an ester linkage. Thus, PC(16:0/22:4(7Z,10Z,13Z,16Z)) is considered to be a glycerophosphocholine lipid molecule. PC(16:0/22:4(7Z,10Z,13Z,16Z)) is a very hydrophobic molecule, practically insoluble (in water), and relatively neutral. PC(16:0/22:4(7Z,10Z,13Z,16Z)) exists in all eukaryotes, ranging from yeast to humans. PC(16:0/22:4(7Z,10Z,13Z,16Z)) participates in a number of enzymatic reactions, within cattle. In particular, S-Adenosylhomocysteine and PC(16:0/22:4(7Z,10Z,13Z,16Z)) can be biosynthesized from S-adenosylmethionine and pe-nme2(16:0/22:4(7Z,10Z,13Z,16Z)) through its interaction with the enzyme phosphatidylethanolamine N-methyltransferase. Furthermore, Cytidine monophosphate and PC(16:0/22:4(7Z,10Z,13Z,16Z)) can be biosynthesized from CDP-choline and DG(16:0/22:4(7Z,10Z,13Z,16Z)/0:0) through the action of the enzyme choline/ethanolaminephosphotransferase. Finally, PC(16:0/22:4(7Z,10Z,13Z,16Z)) and L-serine can be converted into choline and PS(16:0/22:4(7Z,10Z,13Z,16Z)) through the action of the enzyme phosphatidylserine synthase. In cattle, PC(16:0/22:4(7Z,10Z,13Z,16Z)) is involved in a couple of metabolic pathways, which include phosphatidylcholine biosynthesis PC(16:0/22:4(7Z,10Z,13Z,16Z)) pathway and phosphatidylethanolamine biosynthesis pe(16:0/22:4(7Z,10Z,13Z,16Z)) pathway. |
|---|
| Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 1-Hexadecanoyl-2-(7Z,10Z,13Z,16Z-docosatetraenoyl)-sn-glycero-3-phosphocholine | ChEBI | | 1-Palmitoyl-2-adrenoyl-sn-glycero-3-phosphocholine | ChEBI | | GPCho(16:0/22:4) | ChEBI | | GPCho(16:0/22:4n6) | ChEBI | | GPCho(16:0/22:4W6) | ChEBI | | PC(16:0/22:4) | ChEBI | | PC(16:0/22:4n6) | ChEBI | | PC(16:0/22:4W6) | ChEBI | | Phosphatidylcholine(16:0/22:4) | ChEBI | | Phosphatidylcholine(16:0/22:4n6) | ChEBI | | Phosphatidylcholine(16:0/22:4W6) | ChEBI | | PC(38:4) | HMDB | | Phosphatidylcholine(38:4) | HMDB | | GPCho(38:4) | HMDB | | Lecithin | HMDB | | PC(16:0/22:4(7Z,10Z,13Z,16Z)) | Lipid Annotator |
|
|---|
| Chemical Formula | C46H84NO8P |
|---|
| Average Molecular Weight | 810.1348 |
|---|
| Monoisotopic Molecular Weight | 809.593455181 |
|---|
| IUPAC Name | (2-{[(2R)-2-[(7Z,10Z,13Z,16Z)-docosa-7,10,13,16-tetraenoyloxy]-3-(hexadecanoyloxy)propyl phosphonato]oxy}ethyl)trimethylazanium |
|---|
| Traditional Name | lecithin |
|---|
| CAS Registry Number | Not Available |
|---|
| SMILES | CCCCCCCCCCCCCCCC(=O)OC[C@]([H])(COP([O-])(=O)OCC[N+](C)(C)C)OC(=O)CCCCC\C=C/C\C=C/C\C=C/C\C=C/CCCCC |
|---|
| InChI Identifier | InChI=1S/C46H84NO8P/c1-6-8-10-12-14-16-18-20-21-22-23-24-25-27-29-31-33-35-37-39-46(49)55-44(43-54-56(50,51)53-41-40-47(3,4)5)42-52-45(48)38-36-34-32-30-28-26-19-17-15-13-11-9-7-2/h14,16,20-21,23-24,27,29,44H,6-13,15,17-19,22,25-26,28,30-43H2,1-5H3/b16-14-,21-20-,24-23-,29-27-/t44-/m1/s1 |
|---|
| InChI Key | IRWRFKUTKSUFST-MDYGELLQSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as phosphatidylcholines. These are glycerophosphocholines in which the two free -OH are attached to one fatty acid each through an ester linkage. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Lipids and lipid-like molecules |
|---|
| Class | Glycerophospholipids |
|---|
| Sub Class | Glycerophosphocholines |
|---|
| Direct Parent | Phosphatidylcholines |
|---|
| Alternative Parents | |
|---|
| Substituents | - Diacylglycero-3-phosphocholine
- Phosphocholine
- Fatty acid ester
- Dialkyl phosphate
- Dicarboxylic acid or derivatives
- Organic phosphoric acid derivative
- Phosphoric acid ester
- Alkyl phosphate
- Fatty acyl
- Quaternary ammonium salt
- Tetraalkylammonium salt
- Carboxylic acid ester
- Carboxylic acid derivative
- Organic oxygen compound
- Organic nitrogen compound
- Carbonyl group
- Organooxygen compound
- Organonitrogen compound
- Organic oxide
- Organopnictogen compound
- Amine
- Organic salt
- Hydrocarbon derivative
- Aliphatic acyclic compound
|
|---|
| Molecular Framework | Aliphatic acyclic compounds |
|---|
| External Descriptors | |
|---|
| Ontology |
|---|
| Status | Detected and Quantified |
|---|
| Origin | |
|---|
| Biofunction | Not Available |
|---|
| Application | Not Available |
|---|
| Cellular locations | - Cell membrane
- Intracellular membrane
- Membrane
|
|---|
| Physical Properties |
|---|
| State | Solid |
|---|
| Experimental Properties | | Property | Value | Reference |
|---|
| Melting Point | Not Available | Not Available | | Boiling Point | Not Available | Not Available | | Water Solubility | Not Available | Not Available | | LogP | Not Available | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-03di-0000000090-192ed45188cac140e1ed | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-03e9-0600000090-027d9ab618ed6e32122c | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0fai-1900221210-4e9f6502a82e4509b6b1 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-001i-0000000090-bd4db731926b5d2cbe56 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-001i-0000000190-c053420ce1420abe1479 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0002-0900179110-c22771646c354f679e1e | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0006-0000000090-20235538cb799e7cb64e | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0006-0011000090-ac55570378f63c365c9f | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0a66-0099000090-fa15b35ca894ace04e19 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-03di-0000000090-12f7c14a559d8169eb4c | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-03e9-0600000090-4e1c9c4510d5cf1106a3 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0f89-1900221210-cfa97ab46a38175cfaf3 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0a4i-0000000090-fa334b794ce842b59d7b | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0a4i-0057004390-cb754f395b2fc79d4041 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0a4i-7393100000-a5df470b0f09a09ef898 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-014i-0000000090-b25dca7fc1b5373b3809 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-014i-0000000190-58e0c5d29433cc0e7cdf | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-001i-0200498220-16bd6130ed1009a1a231 | View in MoNA |
|---|
|
|---|