| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-10-03 17:14:16 UTC |
|---|
| Update Date | 2020-06-04 20:08:08 UTC |
|---|
| BMDB ID | BMDB0008126 |
|---|
| Secondary Accession Numbers | |
|---|
| Metabolite Identification |
|---|
| Common Name | PC(18:1(9Z)/P-16:0) |
|---|
| Description | PC(18:1(9Z)/P-16:0) is a phosphatidylchloline (PC). It is a glycerophospholipid in which a phosphorylcholine moiety occupies a glycerol substitution site. As is the case with diacylglycerols, phosphatidylcholines can have many different combinations of fatty acids of varying lengths and saturation attached to the C-1 and C-2 positions. PC(18:1(9Z)/P-16:0), in particular, consists of one 9Z-octadecenoyl chain to the C-1 atom, and one 1Z-hexadecenyl to the C-2 atom. In E. coli, PCs can be found in the integral component of the cell outer membrane. They are hydrolyzed by Phospholipases to a 2-acylglycerophosphocholine and a carboxylate. |
|---|
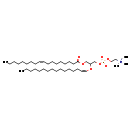
| Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 1-Oleoyl-2-(1-enyl-palmitoyl)-sn-glycero-3-phosphocholine | HMDB | | Gpcho(18:1/16:0) | HMDB | | Gpcho(18:1n9/16:0) | HMDB | | Gpcho(18:1W9/16:0) | HMDB | | Gpcho(34:1) | HMDB | | Lecithin | HMDB | | PC Aa C34:1 | HMDB | | PC(18:1/16:0) | HMDB | | PC(18:1n9/16:0) | HMDB | | PC(18:1W9/16:0) | HMDB | | PC(34:1) | HMDB | | Phosphatidylcholine(18:1/16:0) | HMDB | | Phosphatidylcholine(18:1n9/16:0) | HMDB | | Phosphatidylcholine(18:1W9/16:0) | HMDB | | Phosphatidylcholine(34:1) | HMDB |
|
|---|
| Chemical Formula | C42H82NO7P |
|---|
| Average Molecular Weight | 744.0767 |
|---|
| Monoisotopic Molecular Weight | 743.582890495 |
|---|
| IUPAC Name | [2-({2-[(1Z)-hexadec-1-en-1-yloxy]-3-[(9Z)-octadec-9-enoyloxy]propyl phosphonato}oxy)ethyl]trimethylazanium |
|---|
| Traditional Name | lecithin |
|---|
| CAS Registry Number | Not Available |
|---|
| SMILES | CCCCCCCCCCCCCC\C=C/OC(COC(=O)CCCCCCC\C=C/CCCCCCCC)COP([O-])(=O)OCC[N+](C)(C)C |
|---|
| InChI Identifier | InChI=1S/C42H82NO7P/c1-6-8-10-12-14-16-18-20-22-23-25-27-29-31-33-35-42(44)48-39-41(40-50-51(45,46)49-38-36-43(3,4)5)47-37-34-32-30-28-26-24-21-19-17-15-13-11-9-7-2/h20,22,34,37,41H,6-19,21,23-33,35-36,38-40H2,1-5H3/b22-20-,37-34- |
|---|
| InChI Key | FQVFGIZCMMQDNV-NQQLFREJSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as 1-acyl,2-(1z-alkenyl)-glycerophosphocholines. These are glycerophosphocholines that carry exactly one acyl chain attached to the glycerol moiety through an ester linkage at the O1-position, and one 1Z-alkenyl chain attached through an ether linkage at the O2-position. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Lipids and lipid-like molecules |
|---|
| Class | Glycerophospholipids |
|---|
| Sub Class | Glycerophosphocholines |
|---|
| Direct Parent | 1-acyl,2-(1Z-alkenyl)-glycerophosphocholines |
|---|
| Alternative Parents | |
|---|
| Substituents | - 1-acyl,2-(1z-alkenyl)-glycerophosphocholine
- Phosphocholine
- Glycerol vinyl ether
- Fatty acid ester
- Dialkyl phosphate
- Organic phosphoric acid derivative
- Phosphoric acid ester
- Alkyl phosphate
- Fatty acyl
- Quaternary ammonium salt
- Tetraalkylammonium salt
- Carboxylic acid ester
- Monocarboxylic acid or derivatives
- Carboxylic acid derivative
- Amine
- Organic oxide
- Organopnictogen compound
- Organooxygen compound
- Organonitrogen compound
- Organic oxygen compound
- Organic nitrogen compound
- Carbonyl group
- Hydrocarbon derivative
- Organic salt
- Aliphatic acyclic compound
|
|---|
| Molecular Framework | Aliphatic acyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Ontology |
|---|
| Status | Expected but not Quantified |
|---|
| Origin | |
|---|
| Biofunction | Not Available |
|---|
| Application | Not Available |
|---|
| Cellular locations | - Cell membrane
- Intracellular membrane
- Membrane
|
|---|
| Physical Properties |
|---|
| State | Solid |
|---|
| Experimental Properties | | Property | Value | Reference |
|---|
| Melting Point | Not Available | Not Available | | Boiling Point | Not Available | Not Available | | Water Solubility | Not Available | Not Available | | LogP | Not Available | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | View in JSpectraViewer |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-000i-9060231300-84659a4d58b5b44b90b5 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-02h0-4291111000-a32c8eb5955fa6049c09 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0080-6090022000-efc1ef2b188f27b72438 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-01qc-0090000300-6e9a74ea05c4a3dd5bed | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-01q9-0090001000-6f89e5e3fa02bdf6c3fd | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-01qi-3090100000-9a7173ad1414996fd680 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0006-1100001900-bc2e28c3cd4af881d5b8 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-001i-2900001200-f54c6c5746b6b55cebff | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-001i-1900000000-968591b3ff496ad92785 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0006-0000000900-23872d2e94db2d9fdf31 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0006-0060003900-fda23d09a1d76e6c36c3 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-003r-7091200000-536a61b2f4679ffc2b5d | View in MoNA |
|---|
|
|---|