| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-10-03 18:20:39 UTC |
|---|
| Update Date | 2020-05-11 19:18:30 UTC |
|---|
| BMDB ID | BMDB0011554 |
|---|
| Secondary Accession Numbers | |
|---|
| Metabolite Identification |
|---|
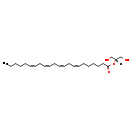
| Common Name | MG(0:0/22:4(7Z,10Z,13Z,16Z)/0:0) |
|---|
| Description | MG(0:0/22:4(7Z,10Z,13Z,16Z)/0:0) belongs to the family of monoradyglycerols, which are glycerolipids lipids containing a common glycerol backbone to which at one fatty acyl group is attached. Their general formula is [R1]OCC(CO[R2])O[R3]. MG(0:0/22:4(7Z,10Z,13Z,16Z)/0:0) is made up of one 7Z,10Z,13Z,16Z-docosatetraenoyl(R2). |
|---|
| Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| Glycerolipid | HMDB | | 2-(7Z,10Z,13Z,16Z-Docosatetraenoyl)-sn-glycerol | HMDB | | Glycerolipid(0:0/22:4(7Z,10Z,13Z,16Z)/0:0) | HMDB | | MG(0:0/22:4/0:0) | HMDB | | MAG(0:0/22:4/0:0) | HMDB | | 1,3-Dihydroxypropan-2-yl (7Z,10Z,13Z,16Z)-docosa-7,10,13,16-tetraenoic acid | HMDB |
|
|---|
| Chemical Formula | C25H42O4 |
|---|
| Average Molecular Weight | 406.5986 |
|---|
| Monoisotopic Molecular Weight | 406.308309832 |
|---|
| IUPAC Name | 1,3-dihydroxypropan-2-yl (7Z,10Z,13Z,16Z)-docosa-7,10,13,16-tetraenoate |
|---|
| Traditional Name | 1,3-dihydroxypropan-2-yl (7Z,10Z,13Z,16Z)-docosa-7,10,13,16-tetraenoate |
|---|
| CAS Registry Number | Not Available |
|---|
| SMILES | [H]C(CO)(CO)OC(=O)CCCCC\C=C/C\C=C/C\C=C/C\C=C/CCCCC |
|---|
| InChI Identifier | InChI=1S/C25H42O4/c1-2-3-4-5-6-7-8-9-10-11-12-13-14-15-16-17-18-19-20-21-25(28)29-24(22-26)23-27/h6-7,9-10,12-13,15-16,24,26-27H,2-5,8,11,14,17-23H2,1H3/b7-6-,10-9-,13-12-,16-15- |
|---|
| InChI Key | DVYDCUGIJCYIQN-DOFZRALJSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as 2-monoacylglycerols. These are monoacylglycerols containing a glycerol acylated at the 2-position. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Lipids and lipid-like molecules |
|---|
| Class | Glycerolipids |
|---|
| Sub Class | Monoradylglycerols |
|---|
| Direct Parent | 2-monoacylglycerols |
|---|
| Alternative Parents | |
|---|
| Substituents | - 2-acyl-sn-glycerol
- Fatty acid ester
- Fatty acyl
- Carboxylic acid ester
- Monocarboxylic acid or derivatives
- Carboxylic acid derivative
- Organic oxygen compound
- Organic oxide
- Hydrocarbon derivative
- Primary alcohol
- Organooxygen compound
- Carbonyl group
- Alcohol
- Aliphatic acyclic compound
|
|---|
| Molecular Framework | Aliphatic acyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Ontology |
|---|
| Status | Expected but not Quantified |
|---|
| Origin | |
|---|
| Biofunction | Not Available |
|---|
| Application | Not Available |
|---|
| Cellular locations | |
|---|
| Physical Properties |
|---|
| State | Solid |
|---|
| Experimental Properties | | Property | Value | Reference |
|---|
| Melting Point | Not Available | Not Available | | Boiling Point | Not Available | Not Available | | Water Solubility | Not Available | Not Available | | LogP | Not Available | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (2 TMS) - 70eV, Positive | splash10-00kr-6292120000-3b3b33f4aa42f5779c9b | View in MoNA |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | View in JSpectraViewer |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-00di-0000900000-f3869cb0507ce31a3515 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0ai0-0000900000-e93c26e489e7f02e4ecb | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-14vi-0004900000-58f56b6978962b47c0fd | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0a4i-0000900000-dd4958aa993a6b51cc58 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0a60-0009700000-f98520e9c01736a55c33 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-00m0-0009000000-013e0d628dd8cebffeaf | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-05g3-9014500000-15711b0a307b2e6a5db6 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-08nc-9005000000-61928a0a2c44de393a56 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0bt9-9205000000-6e24706935a69744451f | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-004i-0000900000-91c15c259057be52fad6 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-004i-0000900000-91c15c259057be52fad6 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0ak9-0009300000-2596b636271078ff6f8e | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-00di-0000900000-5dddac2107ad58eb21f0 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0ab9-0000900000-13cd3bee915e9d020449 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-14i0-0007900000-4045f11a2863a1f6c70a | View in MoNA |
|---|
|
|---|