| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-10-03 18:23:51 UTC |
|---|
| Update Date | 2020-05-05 18:38:32 UTC |
|---|
| BMDB ID | BMDB0011741 |
|---|
| Secondary Accession Numbers | |
|---|
| Metabolite Identification |
|---|
| Common Name | Gamma-Glutamyltyrosine |
|---|
| Description | gamma-Glutamyltyrosine, also known as γ-L-glutamyl-L-tyrosine or γ-glu-tyr, belongs to the class of organic compounds known as tyrosine and derivatives. Tyrosine and derivatives are compounds containing tyrosine or a derivative thereof resulting from reaction of tyrosine at the amino group or the carboxy group, or from the replacement of any hydrogen of glycine by a heteroatom. Based on a literature review a significant number of articles have been published on gamma-Glutamyltyrosine. |
|---|
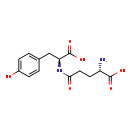
| Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| g-Glutamyltyrosine | Generator | | Γ-glutamyltyrosine | Generator | | Γ-glu-tyr | HMDB | | Γ-L-glu-L-tyr | HMDB | | Γ-L-glutamyl-L-tyrosine | HMDB | | L-Γ-glutamyl-L-tyrosine | HMDB | | N-Γ-glutamyltyrosine | HMDB | | N-L-Γ-glutamyltyrosine | HMDB | | N-L-Γ-glutamyl-L-tyrosine | HMDB | | gamma-Glu-tyr | HMDB | | gamma-L-Glu-L-tyr | HMDB | | gamma-L-Glutamyl-L-tyrosine | HMDB | | L-gamma-Glutamyl-L-tyrosine | HMDB | | N-gamma-Glutamyltyrosine | HMDB | | N-L-gamma-Glutamyltyrosine | HMDB | | N-L-gamma-Glutamyl-L-tyrosine | HMDB | | gamma-Glutamyl-L-tyrosine | HMDB | | N-Γ-L-glutamyl-L-tyrosine | HMDB | | N-gamma-L-Glutamyl-L-tyrosine | HMDB | | g-Glu-tyr | HMDB | | gamma-Glutamyltyrosine | HMDB, ChEBI |
|
|---|
| Chemical Formula | C14H18N2O6 |
|---|
| Average Molecular Weight | 310.3025 |
|---|
| Monoisotopic Molecular Weight | 310.116486318 |
|---|
| IUPAC Name | (2S)-2-amino-4-{[(1S)-1-carboxy-2-(4-hydroxyphenyl)ethyl]carbamoyl}butanoic acid |
|---|
| Traditional Name | (2S)-2-amino-4-{[(1S)-1-carboxy-2-(4-hydroxyphenyl)ethyl]carbamoyl}butanoic acid |
|---|
| CAS Registry Number | 7432-23-7 |
|---|
| SMILES | N[C@@H](CCC(=O)N[C@@H](CC1=CC=C(O)C=C1)C(O)=O)C(O)=O |
|---|
| InChI Identifier | InChI=1S/C14H18N2O6/c15-10(13(19)20)5-6-12(18)16-11(14(21)22)7-8-1-3-9(17)4-2-8/h1-4,10-11,17H,5-7,15H2,(H,16,18)(H,19,20)(H,21,22)/t10-,11-/m0/s1 |
|---|
| InChI Key | VVLXCWVSSLFQDS-QWRGUYRKSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as tyrosine and derivatives. Tyrosine and derivatives are compounds containing tyrosine or a derivative thereof resulting from reaction of tyrosine at the amino group or the carboxy group, or from the replacement of any hydrogen of glycine by a heteroatom. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organic acids and derivatives |
|---|
| Class | Carboxylic acids and derivatives |
|---|
| Sub Class | Amino acids, peptides, and analogues |
|---|
| Direct Parent | Tyrosine and derivatives |
|---|
| Alternative Parents | |
|---|
| Substituents | - Tyrosine or derivatives
- Phenylalanine or derivatives
- N-acyl-alpha-amino acid
- N-acyl-alpha amino acid or derivatives
- 3-phenylpropanoic-acid
- Alpha-amino acid
- Amphetamine or derivatives
- L-alpha-amino acid
- 1-hydroxy-2-unsubstituted benzenoid
- Phenol
- Monocyclic benzene moiety
- Dicarboxylic acid or derivatives
- Benzenoid
- Amino acid
- Carboximidic acid
- Carboximidic acid derivative
- Carboxylic acid
- Organic 1,3-dipolar compound
- Propargyl-type 1,3-dipolar organic compound
- Hydrocarbon derivative
- Primary aliphatic amine
- Organic oxide
- Organic oxygen compound
- Amine
- Organic nitrogen compound
- Carbonyl group
- Organonitrogen compound
- Organooxygen compound
- Organopnictogen compound
- Primary amine
- Aromatic homomonocyclic compound
|
|---|
| Molecular Framework | Aromatic homomonocyclic compounds |
|---|
| External Descriptors | |
|---|
| Ontology |
|---|
| Status | Detected but not Quantified |
|---|
| Origin | |
|---|
| Biofunction | Not Available |
|---|
| Application | Not Available |
|---|
| Cellular locations | Not Available |
|---|
| Physical Properties |
|---|
| State | Solid |
|---|
| Experimental Properties | | Property | Value | Reference |
|---|
| Melting Point | Not Available | Not Available | | Boiling Point | Not Available | Not Available | | Water Solubility | Not Available | Not Available | | LogP | Not Available | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-066u-8970000000-7698a5449443ffa93dd7 | View in MoNA |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (3 TMS) - 70eV, Positive | splash10-03k9-7943780000-b133994d06ffbb64bbc5 | View in MoNA |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | View in JSpectraViewer |
|---|
| LC-MS/MS | LC-MS/MS Spectrum - LC-ESI-IT , positive | splash10-001i-0900000000-6ab9af8fad2893eaf075 | View in MoNA |
|---|
| LC-MS/MS | LC-MS/MS Spectrum - 35V, Positive | splash10-015i-0900000000-d7175d83339cd2872759 | View in MoNA |
|---|
| LC-MS/MS | LC-MS/MS Spectrum - 35V, Negative | splash10-004i-0900000000-4592035b078ebbd2cb85 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-02tc-0491000000-4cae131578a3279f443d | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0159-2890000000-0478e3d3eadc801de363 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0a4i-9810000000-9784da266f6bbea3de28 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0a4i-0197000000-8b8dfc31e6120620b031 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-05ng-1791000000-581438da50874618543b | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-00ec-7900000000-b290dc16b839c2237741 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-06sl-1953000000-a2d27d1829e1d59b4f96 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-02ji-2920000000-1b9344a2e5412bdbc2f3 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-00dl-9600000000-86b4704920a23ca4164e | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-03yr-1975000000-f9c07a27e39547704d3b | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0019-2910000000-b220ff5f3228fb1b2d79 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0a59-9400000000-af3a54518d3a4933e229 | View in MoNA |
|---|
| 1D NMR | 1H NMR Spectrum (1D, 100 MHz, D2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 13C NMR Spectrum (1D, 100 MHz, D2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 1H NMR Spectrum (1D, 1000 MHz, D2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 13C NMR Spectrum (1D, 1000 MHz, D2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 1H NMR Spectrum (1D, 200 MHz, D2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 13C NMR Spectrum (1D, 200 MHz, D2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 1H NMR Spectrum (1D, 300 MHz, D2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 13C NMR Spectrum (1D, 300 MHz, D2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 1H NMR Spectrum (1D, 400 MHz, D2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 13C NMR Spectrum (1D, 400 MHz, D2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 1H NMR Spectrum (1D, 500 MHz, D2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 13C NMR Spectrum (1D, 500 MHz, D2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 1H NMR Spectrum (1D, 600 MHz, D2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 13C NMR Spectrum (1D, 600 MHz, D2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 1H NMR Spectrum (1D, 700 MHz, D2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 13C NMR Spectrum (1D, 700 MHz, D2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 1H NMR Spectrum (1D, 800 MHz, D2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 13C NMR Spectrum (1D, 800 MHz, D2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 1H NMR Spectrum (1D, 900 MHz, D2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 13C NMR Spectrum (1D, 900 MHz, D2O, predicted) | Not Available | View in JSpectraViewer |
|---|
|
|---|