| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2018-06-25 21:57:34 UTC |
|---|
| Update Date | 2020-05-05 18:40:40 UTC |
|---|
| BMDB ID | BMDB0062195 |
|---|
| Secondary Accession Numbers | |
|---|
| Metabolite Identification |
|---|
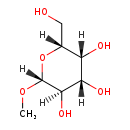
| Common Name | Methyl beta-D-glucopyranoside |
|---|
| Description | Methyl beta-galactoside, also known as galbetaome, belongs to the class of organic compounds known as o-glycosyl compounds. These are glycoside in which a sugar group is bonded through one carbon to another group via a O-glycosidic bond. Methyl beta-galactoside is an extremely weak basic (essentially neutral) compound (based on its pKa). |
|---|
| Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| beta-D-Galactopyranose methyl glycoside | ChEBI | | GalbetaOMe | ChEBI | | Methyl beta-D-galactopyranoside | ChEBI | | Methyl galactoside | ChEBI | | Methylgalactoside | ChEBI | | b-D-Galactopyranose methyl glycoside | Generator | | Β-D-galactopyranose methyl glycoside | Generator | | Methyl b-D-galactopyranoside | Generator | | Methyl β-D-galactopyranoside | Generator | | Methyl b-galactoside | Generator | | Methyl β-galactoside | Generator | | Methyl beta-galactoside | ChEBI | | Methyl-b-D-galactoside | Generator | | Methyl-β-D-galactoside | Generator | | beta-O-Methyl-galactopyranoside | MeSH | | Methyl beta-galactoside, (alpha-L)-isomer | MeSH | | beta-O-Methyl-D-galactopyranoside | MeSH | | Methyl-beta-D-galactopyranoside | MeSH | | Methyl-beta-galactopyranoside | MeSH | | 3-O-Methyl-D-galactose | MeSH | | Methyl beta-galactoside, (alpha-D)-isomer | MeSH | | Methyl beta-galactoside, (beta-L)-isomer | MeSH | | Methyl b-D-glucopyranoside | Generator | | Methyl β-D-glucopyranoside | Generator |
|
|---|
| Chemical Formula | C7H14O6 |
|---|
| Average Molecular Weight | 194.1825 |
|---|
| Monoisotopic Molecular Weight | 194.07903818 |
|---|
| IUPAC Name | (2R,3R,4S,5R,6R)-2-(hydroxymethyl)-6-methoxyoxane-3,4,5-triol |
|---|
| Traditional Name | methyl galactoside |
|---|
| CAS Registry Number | 1824-94-8 |
|---|
| SMILES | [H][C@]1(O)[C@@]([H])(O)[C@@]([H])(CO)O[C@@]([H])(OC)[C@]1([H])O |
|---|
| InChI Identifier | InChI=1S/C7H14O6/c1-12-7-6(11)5(10)4(9)3(2-8)13-7/h3-11H,2H2,1H3/t3-,4+,5+,6-,7-/m1/s1 |
|---|
| InChI Key | HOVAGTYPODGVJG-VOQCIKJUSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as o-glycosyl compounds. These are glycoside in which a sugar group is bonded through one carbon to another group via a O-glycosidic bond. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organic oxygen compounds |
|---|
| Class | Organooxygen compounds |
|---|
| Sub Class | Carbohydrates and carbohydrate conjugates |
|---|
| Direct Parent | O-glycosyl compounds |
|---|
| Alternative Parents | |
|---|
| Substituents | - O-glycosyl compound
- Oxane
- Monosaccharide
- Secondary alcohol
- Oxacycle
- Organoheterocyclic compound
- Polyol
- Acetal
- Hydrocarbon derivative
- Primary alcohol
- Alcohol
- Aliphatic heteromonocyclic compound
|
|---|
| Molecular Framework | Aliphatic heteromonocyclic compounds |
|---|
| External Descriptors | |
|---|
| Ontology |
|---|
| Status | Detected but not Quantified |
|---|
| Origin | |
|---|
| Biofunction | Not Available |
|---|
| Application | Not Available |
|---|
| Cellular locations | |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Experimental Properties | | Property | Value | Reference |
|---|
| Melting Point | Not Available | Not Available | | Boiling Point | Not Available | Not Available | | Water Solubility | Not Available | Not Available | | LogP | Not Available | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | |
|---|
| GC-MS | GC-MS Spectrum - GC-MS (4 TMS) | splash10-0ue9-1980000000-f85f2b643334aeddd3a5 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-002b-0900000000-e857ad38620167398499 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-004j-3900000000-72598960d60412f0f056 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0btd-9300000000-61f6e6dd66385e4c3845 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0006-1900000000-6c0b4ecf5c8009ef4282 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0096-6900000000-196e3ebd8371553bafba | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-006x-9100000000-47c83236e172e9d82eac | View in MoNA |
|---|
| 1D NMR | 13C NMR Spectrum (1D, 100 MHz, H2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 1H NMR Spectrum (1D, 100 MHz, H2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 13C NMR Spectrum (1D, 1000 MHz, H2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 1H NMR Spectrum (1D, 1000 MHz, H2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 13C NMR Spectrum (1D, 200 MHz, H2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 1H NMR Spectrum (1D, 200 MHz, H2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 13C NMR Spectrum (1D, 300 MHz, H2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 1H NMR Spectrum (1D, 300 MHz, H2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 13C NMR Spectrum (1D, 400 MHz, H2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 1H NMR Spectrum (1D, 400 MHz, H2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 13C NMR Spectrum (1D, 500 MHz, H2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 1H NMR Spectrum (1D, 500 MHz, H2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 13C NMR Spectrum (1D, 600 MHz, H2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 1H NMR Spectrum (1D, 600 MHz, H2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 13C NMR Spectrum (1D, 700 MHz, H2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 1H NMR Spectrum (1D, 700 MHz, H2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 13C NMR Spectrum (1D, 800 MHz, H2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 1H NMR Spectrum (1D, 800 MHz, H2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 13C NMR Spectrum (1D, 900 MHz, H2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 1H NMR Spectrum (1D, 900 MHz, H2O, predicted) | Not Available | View in JSpectraViewer |
|---|
|
|---|
| General References | - Melzer N, Wittenburg D, Hartwig S, Jakubowski S, Kesting U, Willmitzer L, Lisec J, Reinsch N, Repsilber D: Investigating associations between milk metabolite profiles and milk traits of Holstein cows. J Dairy Sci. 2013 Mar;96(3):1521-34. doi: 10.3168/jds.2012-5743. [PubMed:23438684 ]
|
|---|