| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2018-08-29 17:17:05 UTC |
|---|
| Update Date | 2020-06-04 20:30:02 UTC |
|---|
| BMDB ID | BMDB0063125 |
|---|
| Secondary Accession Numbers | |
|---|
| Metabolite Identification |
|---|
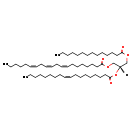
| Common Name | TG(14:0/18:1(9Z)/20:3(8Z,11Z,14Z))[iso6] |
|---|
| Description | TG(14:0/18:1(9Z)/20:3(8Z,11Z,14Z))[iso6] belongs to the class of organic compounds known as triacylglycerols. These are glycerides consisting of three fatty acid chains covalently bonded to a glycerol molecule through ester linkages. Thus, TG(14:0/18:1(9Z)/20:3(8Z,11Z,14Z))[iso6] is considered to be a triradylglycerol lipid molecule. TG(14:0/18:1(9Z)/20:3(8Z,11Z,14Z))[iso6] is a very hydrophobic molecule, practically insoluble (in water), and relatively neutral. |
|---|
| Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 1-Myristoyl-2-oleoyl-3-homo-g-linolenoyl-glycerol | HMDB | | 1-Tetradecanoyl-2-(9Z-octadecenoyl)-3-(8Z,11Z,14Z-eicosatrienoyl)-glycerol | HMDB | | TAG(14:0/18:1/20:3) | HMDB | | TAG(14:0/18:1/20:3n6) | HMDB | | TAG(14:0/18:1/20:3W6) | HMDB | | TAG(52:4) | HMDB | | TG(14:0/18:1/20:3) | HMDB | | TG(14:0/18:1/20:3n6) | HMDB | | TG(14:0/18:1/20:3W6) | HMDB | | TG(52:4) | HMDB | | Tracylglycerol(14:0/18:1/20:3) | HMDB | | Tracylglycerol(14:0/18:1/20:3n6) | HMDB | | Tracylglycerol(14:0/18:1/20:3W6) | HMDB | | Tracylglycerol(52:4) | HMDB | | Triacylglycerol | HMDB | | Triglyceride | HMDB | | 1-Myristoyl-2-oleoyl-3-dihomo-gamma-linolenoyl-glycerol | HMDB | | TG(14:0/18:1n9/20:3n6) | HMDB | | TG(14:0/18:1W9/20:3W6) | HMDB | | Tag(14:0/18:1(9Z)/20:3(8Z,11Z,14Z)) | HMDB | | Tag(14:0/18:1n9/20:3n6) | HMDB | | Tag(14:0/18:1W9/20:3W6) | HMDB | | Triacylglycerol(14:0/18:1(9Z)/20:3(8Z,11Z,14Z)) | HMDB | | Triacylglycerol(14:0/18:1/20:3) | HMDB | | Triacylglycerol(14:0/18:1n9/20:3n6) | HMDB | | Triacylglycerol(14:0/18:1W9/20:3W6) | HMDB | | Triacylglycerol(52:4) | HMDB | | TG(14:0/18:1(9Z)/20:3(8Z,11Z,14Z)) | HMDB | | TG(14:0/18:1(9Z)/20:3n6) | Lipid Annotator |
|
|---|
| Chemical Formula | C55H98O6 |
|---|
| Average Molecular Weight | 855.383 |
|---|
| Monoisotopic Molecular Weight | 854.736340876 |
|---|
| IUPAC Name | (2S)-2-[(9Z)-octadec-9-enoyloxy]-3-(tetradecanoyloxy)propyl (8Z,11Z,14Z)-icosa-8,11,14-trienoate |
|---|
| Traditional Name | (2S)-2-[(9Z)-octadec-9-enoyloxy]-3-(tetradecanoyloxy)propyl (8Z,11Z,14Z)-icosa-8,11,14-trienoate |
|---|
| CAS Registry Number | Not Available |
|---|
| SMILES | [H][C@](COC(=O)CCCCCCCCCCCCC)(COC(=O)CCCCCC\C=C/C\C=C/C\C=C/CCCCC)OC(=O)CCCCCCC\C=C/CCCCCCCC |
|---|
| InChI Identifier | InChI=1S/C55H98O6/c1-4-7-10-13-16-19-22-24-26-27-29-30-33-36-39-42-45-48-54(57)60-51-52(50-59-53(56)47-44-41-38-35-32-21-18-15-12-9-6-3)61-55(58)49-46-43-40-37-34-31-28-25-23-20-17-14-11-8-5-2/h16,19,24-26,28-30,52H,4-15,17-18,20-23,27,31-51H2,1-3H3/b19-16-,26-24-,28-25-,30-29-/t52-/m0/s1 |
|---|
| InChI Key | NUCVTMFYAFILJA-YGWJPMFMSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as triacylglycerols. These are glycerides consisting of three fatty acid chains covalently bonded to a glycerol molecule through ester linkages. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Lipids and lipid-like molecules |
|---|
| Class | Glycerolipids |
|---|
| Sub Class | Triradylcglycerols |
|---|
| Direct Parent | Triacylglycerols |
|---|
| Alternative Parents | |
|---|
| Substituents | - Triacyl-sn-glycerol
- Tricarboxylic acid or derivatives
- Fatty acid ester
- Fatty acyl
- Carboxylic acid ester
- Carboxylic acid derivative
- Organic oxygen compound
- Organic oxide
- Hydrocarbon derivative
- Organooxygen compound
- Carbonyl group
- Aliphatic acyclic compound
|
|---|
| Molecular Framework | Aliphatic acyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Ontology |
|---|
| Status | Detected and Quantified |
|---|
| Origin | Not Available |
|---|
| Biofunction | Not Available |
|---|
| Application | Not Available |
|---|
| Cellular locations | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Experimental Properties | | Property | Value | Reference |
|---|
| Melting Point | Not Available | Not Available | | Boiling Point | Not Available | Not Available | | Water Solubility | Not Available | Not Available | | LogP | Not Available | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0a70-0092010010-15d56c3c9f44d9965dfd | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0a6r-0094000000-7c7dfd3b988750f8c58e | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0a6r-2092000000-e680d3ffe9a7c50f0625 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0zi1-0097082070-fca31cb9f1bc5c1e7b9c | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0a4j-0059000000-309dfecad5cfbdfbe86c | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0a4i-3089000000-bac9b0fce480b7572250 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-00di-0000000090-6208a1c55097185c43bc | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-00di-0000000090-6208a1c55097185c43bc | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-05i1-0000094030-5161801354720f8b7a88 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-03di-0000000090-59539dfb428f41ddecf8 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-03di-0000000090-59539dfb428f41ddecf8 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-03gi-0009099090-f87c958eee092324a95e | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-00di-0000000090-41f870198c6bd847264f | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-00di-0000000090-41f870198c6bd847264f | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-05i1-0020094030-6eb30080d7ba428491e9 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-004i-0000000090-336a6e861e77b5821f30 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-004i-0000000090-336a6e861e77b5821f30 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-004i-0000000090-336a6e861e77b5821f30 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0a4i-4250041290-df66bf36a39802351284 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-014i-4291001640-4205c900eef71981ba9b | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-014j-1596010200-5766ae72bb85877a65c9 | View in MoNA |
|---|
|
|---|