| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2020-03-10 16:59:10 UTC |
|---|
| Update Date | 2020-04-22 18:55:17 UTC |
|---|
| BMDB ID | BMDB0095976 |
|---|
| Secondary Accession Numbers | |
|---|
| Metabolite Identification |
|---|
| Common Name | Dynorphin B (10-13) |
|---|
| Description | Dynorphin B (10-13) belongs to the class of organic compounds known as oligopeptides. These are organic compounds containing a sequence of between three and ten alpha-amino acids joined by peptide bonds. Based on a literature review very few articles have been published on Dynorphin B (10-13). |
|---|
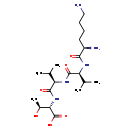
| Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| Lys-val-val-THR | HMDB | | (2S,3S)-2-{[(2R)-2-{[(2S)-2-{[(2R)-2,6-diamino-1-hydroxyhexylidene]amino}-1-hydroxy-3-methylbutylidene]amino}-1-hydroxy-3-methylbutylidene]amino}-3-hydroxybutanoate | Generator |
|
|---|
| Chemical Formula | C20H39N5O6 |
|---|
| Average Molecular Weight | 445.5536 |
|---|
| Monoisotopic Molecular Weight | 445.290034005 |
|---|
| IUPAC Name | (2S,3S)-2-[(2R)-2-[(2S)-2-[(2R)-2,6-diaminohexanamido]-3-methylbutanamido]-3-methylbutanamido]-3-hydroxybutanoic acid |
|---|
| Traditional Name | (2S,3S)-2-[(2R)-2-[(2S)-2-[(2R)-2,6-diaminohexanamido]-3-methylbutanamido]-3-methylbutanamido]-3-hydroxybutanoic acid |
|---|
| CAS Registry Number | Not Available |
|---|
| SMILES | CC(C)[C@H](NC(=O)[C@H](N)CCCCN)C(=O)N[C@H](C(C)C)C(=O)N[C@@H]([C@H](C)O)C(O)=O |
|---|
| InChI Identifier | InChI=1S/C20H39N5O6/c1-10(2)14(23-17(27)13(22)8-6-7-9-21)18(28)24-15(11(3)4)19(29)25-16(12(5)26)20(30)31/h10-16,26H,6-9,21-22H2,1-5H3,(H,23,27)(H,24,28)(H,25,29)(H,30,31)/t12-,13+,14-,15+,16-/m0/s1 |
|---|
| InChI Key | VSLCIGXQLCYQTD-NPJQDHAYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as oligopeptides. These are organic compounds containing a sequence of between three and ten alpha-amino acids joined by peptide bonds. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organic acids and derivatives |
|---|
| Class | Carboxylic acids and derivatives |
|---|
| Sub Class | Amino acids, peptides, and analogues |
|---|
| Direct Parent | Oligopeptides |
|---|
| Alternative Parents | |
|---|
| Substituents | - Alpha-oligopeptide
- Valine or derivatives
- N-acyl-l-alpha-amino acid
- N-acyl-alpha-amino acid
- N-acyl-alpha amino acid or derivatives
- Alpha-amino acid amide
- N-substituted-alpha-amino acid
- Alpha-amino acid or derivatives
- Beta-hydroxy acid
- Branched fatty acid
- Hydroxy fatty acid
- Short-chain hydroxy acid
- Fatty amide
- Hydroxy acid
- N-acyl-amine
- Fatty acyl
- Fatty acid
- Secondary carboxylic acid amide
- Secondary alcohol
- Amino acid or derivatives
- Amino acid
- Carboxamide group
- Monocarboxylic acid or derivatives
- Carboxylic acid
- Organopnictogen compound
- Alcohol
- Hydrocarbon derivative
- Carbonyl group
- Primary aliphatic amine
- Organic oxide
- Organic nitrogen compound
- Amine
- Organonitrogen compound
- Organooxygen compound
- Primary amine
- Organic oxygen compound
- Aliphatic acyclic compound
|
|---|
| Molecular Framework | Aliphatic acyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Ontology |
|---|
| Status | Expected but not Quantified |
|---|
| Origin | |
|---|
| Biofunction | Not Available |
|---|
| Application | Not Available |
|---|
| Cellular locations | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Experimental Properties | | Property | Value | Reference |
|---|
| Melting Point | Not Available | Not Available | | Boiling Point | Not Available | Not Available | | Water Solubility | Not Available | Not Available | | LogP | Not Available | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-0udi-9701300000-f6d8e6dc80bf5b6d3d8e | View in MoNA |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (2 TMS) - 70eV, Positive | splash10-0udi-7900010000-e490c635ccb215159183 | View in MoNA |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | View in JSpectraViewer |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0fb9-0431900000-95699ec60394d252b529 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0udi-6952200000-5286410d2bfca8b4be73 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0fk9-9510000000-a62b49f7fdfbb89beb42 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0udl-0123900000-abe0254fd811d6dac3bd | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0udi-2779700000-68dc2ab4068753d85597 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0g4i-4940000000-820302d9503335e42c7a | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-004l-0001900000-03ff5ba75bc622867440 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0f96-0895700000-4a5f849772038a29cf54 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0kbf-7910000000-6854d3a0eb8e9a376ad1 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0002-0131900000-0894112b9dbdcfa7a085 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0uxs-5796500000-3e9ea816bcaa974dca14 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0udi-7920000000-e14ba1a981dab2fc97c4 | View in MoNA |
|---|
| 1D NMR | 13C NMR Spectrum (1D, 100 MHz, D2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 1H NMR Spectrum (1D, 100 MHz, D2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 13C NMR Spectrum (1D, 1000 MHz, D2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 1H NMR Spectrum (1D, 1000 MHz, D2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 13C NMR Spectrum (1D, 200 MHz, D2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 1H NMR Spectrum (1D, 200 MHz, D2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 13C NMR Spectrum (1D, 300 MHz, D2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 1H NMR Spectrum (1D, 300 MHz, D2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 13C NMR Spectrum (1D, 400 MHz, D2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 1H NMR Spectrum (1D, 400 MHz, D2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 13C NMR Spectrum (1D, 500 MHz, D2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 1H NMR Spectrum (1D, 500 MHz, D2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 13C NMR Spectrum (1D, 600 MHz, D2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 1H NMR Spectrum (1D, 600 MHz, D2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 13C NMR Spectrum (1D, 700 MHz, D2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 1H NMR Spectrum (1D, 700 MHz, D2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 13C NMR Spectrum (1D, 800 MHz, D2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 1H NMR Spectrum (1D, 800 MHz, D2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 13C NMR Spectrum (1D, 900 MHz, D2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 1H NMR Spectrum (1D, 900 MHz, D2O, predicted) | Not Available | View in JSpectraViewer |
|---|
|
|---|