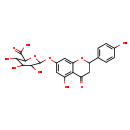| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2020-03-10 17:01:27 UTC |
|---|
| Update Date | 2020-05-11 20:27:26 UTC |
|---|
| BMDB ID | BMDB0096115 |
|---|
| Secondary Accession Numbers | |
|---|
| Metabolite Identification |
|---|
| Common Name | naringenin-7-O-glucuronide |
|---|
| Description | naringenin-7-O-glucuronide belongs to the class of organic compounds known as flavonoid-7-o-glucuronides. These are phenolic compounds containing a flavonoid moiety which is O-glycosidically linked to glucuronic acid at the C7-position. Based on a literature review very few articles have been published on naringenin-7-O-glucuronide. |
|---|
| Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 3,4,5-Trihydroxy-6-{[5-hydroxy-2-(4-hydroxyphenyl)-4-oxo-3,4-dihydro-2H-1-benzopyran-7-yl]oxy}oxane-2-carboxylate | HMDB |
|
|---|
| Chemical Formula | C21H20O11 |
|---|
| Average Molecular Weight | 448.3769 |
|---|
| Monoisotopic Molecular Weight | 448.100561482 |
|---|
| IUPAC Name | 3,4,5-trihydroxy-6-{[5-hydroxy-2-(4-hydroxyphenyl)-4-oxo-3,4-dihydro-2H-1-benzopyran-7-yl]oxy}oxane-2-carboxylic acid |
|---|
| Traditional Name | 3,4,5-trihydroxy-6-{[5-hydroxy-2-(4-hydroxyphenyl)-4-oxo-2,3-dihydro-1-benzopyran-7-yl]oxy}oxane-2-carboxylic acid |
|---|
| CAS Registry Number | Not Available |
|---|
| SMILES | OC1C(O)C(OC2=CC(O)=C3C(=O)CC(OC3=C2)C2=CC=C(O)C=C2)OC(C1O)C(O)=O |
|---|
| InChI Identifier | InChI=1S/C21H20O11/c22-9-3-1-8(2-4-9)13-7-12(24)15-11(23)5-10(6-14(15)31-13)30-21-18(27)16(25)17(26)19(32-21)20(28)29/h1-6,13,16-19,21-23,25-27H,7H2,(H,28,29) |
|---|
| InChI Key | BDCRTIDKZGEVEN-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as flavonoid-7-o-glucuronides. These are phenolic compounds containing a flavonoid moiety which is O-glycosidically linked to glucuronic acid at the C7-position. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Phenylpropanoids and polyketides |
|---|
| Class | Flavonoids |
|---|
| Sub Class | Flavonoid glycosides |
|---|
| Direct Parent | Flavonoid-7-O-glucuronides |
|---|
| Alternative Parents | |
|---|
| Substituents | - Flavonoid-7-o-glucuronide
- Flavonoid-7-o-glycoside
- 4'-hydroxyflavonoid
- 5-hydroxyflavonoid
- Hydroxyflavonoid
- Flavanone
- Flavan
- Phenolic glycoside
- 1-o-glucuronide
- O-glucuronide
- Glucuronic acid or derivatives
- O-glycosyl compound
- Glycosyl compound
- Chromone
- 1-benzopyran
- Benzopyran
- Chromane
- Aryl alkyl ketone
- Aryl ketone
- Beta-hydroxy acid
- Phenol
- 1-hydroxy-2-unsubstituted benzenoid
- Alkyl aryl ether
- 1-hydroxy-4-unsubstituted benzenoid
- Monocyclic benzene moiety
- Benzenoid
- Pyran
- Oxane
- Hydroxy acid
- Monosaccharide
- Vinylogous acid
- Secondary alcohol
- Ketone
- Polyol
- Acetal
- Ether
- Monocarboxylic acid or derivatives
- Carboxylic acid
- Carboxylic acid derivative
- Organoheterocyclic compound
- Oxacycle
- Organic oxide
- Organic oxygen compound
- Alcohol
- Hydrocarbon derivative
- Organooxygen compound
- Carbonyl group
- Aromatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aromatic heteropolycyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Ontology |
|---|
| Status | Expected but not Quantified |
|---|
| Origin | |
|---|
| Biofunction | Not Available |
|---|
| Application | Not Available |
|---|
| Cellular locations | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Experimental Properties | | Property | Value | Reference |
|---|
| Melting Point | Not Available | Not Available | | Boiling Point | Not Available | Not Available | | Water Solubility | Not Available | Not Available | | LogP | Not Available | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-0bu0-9115200000-27930d7f2fd2eddf6aac | View in MoNA |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (3 TMS) - 70eV, Positive | splash10-0f6t-5430339000-8a698e9e4ff6a40f95b8 | View in MoNA |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | View in JSpectraViewer |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-00ea-0291700000-d64b93cbf7380fcecddd | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-05fr-0490100000-683c27201db7f337e2b8 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0pi9-1950000000-1f0e65f8fca829ed6814 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0fdk-2251900000-62c0f368cb0dc57c7e85 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-00di-1291200000-547fccb933ca5a185dc4 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-00di-4490000000-09c6284c98a6b41b69c8 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0002-0030900000-9099ae6cceec31e27e6e | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-00dj-0190700000-32bd1c30a5eaea8d8881 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0fk9-0970200000-59bc4407439899017b5f | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-00dj-0090500000-01e33ad94c06b74ac17b | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-00di-0190000000-5be014833fa610089e5b | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0fk9-0890000000-2b7570a316bc5cbaab9f | View in MoNA |
|---|
|
|---|