| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-09-30 22:18:41 UTC |
|---|
| Update Date | 2020-05-11 20:54:33 UTC |
|---|
| BMDB ID | BMDB0000031 |
|---|
| Secondary Accession Numbers | |
|---|
| Metabolite Identification |
|---|
| Common Name | Androsterone |
|---|
| Description | Androsterone, also known as androkinine or atromide ici, belongs to the class of organic compounds known as androgens and derivatives. These are 3-hydroxylated C19 steroid hormones. They are known to favor the development of masculine characteristics. They also show profound effects on scalp and body hair in humans. Thus, androsterone is considered to be a steroid lipid molecule. Androsterone is a very hydrophobic molecule, practically insoluble (in water), and relatively neutral. |
|---|
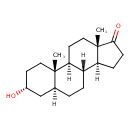
| Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| (3alpha,5alpha)-3-Hydroxyandrostan-17-one | ChEBI | | 3-Epihydroxyetioallocholan-17-one | ChEBI | | 3alpha-Hydroxy-5alpha-androstan-17-one | ChEBI | | 3alpha-Hydroxyetioallocholan-17-one | ChEBI | | 5alpha-Androstane-3alpha-ol-17-one | ChEBI | | 5alpha-Androsterone | ChEBI | | Androkinine | ChEBI | | Androtine | ChEBI | | cis-Androsterone | ChEBI | | (3a,5a)-3-Hydroxyandrostan-17-one | Generator | | (3Α,5α)-3-hydroxyandrostan-17-one | Generator | | 3a-Hydroxy-5a-androstan-17-one | Generator | | 3Α-hydroxy-5α-androstan-17-one | Generator | | 3a-Hydroxyetioallocholan-17-one | Generator | | 3Α-hydroxyetioallocholan-17-one | Generator | | 5a-Androstane-3a-ol-17-one | Generator | | 5Α-androstane-3α-ol-17-one | Generator | | 5a-Androsterone | Generator | | 5Α-androsterone | Generator | | 3-alpha-Hydroxy-5-alpha-androstan-17-one | HMDB | | 3 alpha Hydroxy 5 alpha androstan 17 one | HMDB | | 5 alpha Androstan 3 alpha ol 17 one | HMDB | | 5 alpha-Androstan-3 alpha-ol-17-one | HMDB | | Epiandrosterone | HMDB | | 3-alpha-Hydroxy-17-androstanone | HMDB | | 3-alpha-Hydroxy-5-alpha-androstane-17-one | HMDB | | 3-alpha-Hydroxy-5alpha-androstan-17-one | HMDB | | 3-alpha-Hydroxyetioallocholan-17-one | HMDB | | 3-Hydroxy-(3-alpha,5-alpha)-androstan-17-one | HMDB | | 3-Hydroxyandrostan-17-one | HMDB | | 3alpha-Hydroxy-17-androstanone | HMDB | | 5-alpha-Androstan-3-alpha-ol-17-one | HMDB | | 5-alpha-Androstane-3alpha-ol-17-one | HMDB | | 5-alpha-Androsterone | HMDB | | Androstanon-3-alpha-ol-17-one | HMDB | | Atromide ici | HMDB | | 3Α-hydroxy-17-androstanone | HMDB | | 3Α-hydroxy-5α-androstane-17-one | HMDB | | 5Α-androstan-17-one-3α-ol | HMDB | | 5Α-androstan-3α-ol-17-one | HMDB | | 3alpha-Hydroxy-5alpha-androstane-17-one | HMDB | | 5alpha-Androstan-17-one-3alpha-ol | HMDB | | 5alpha-Androstan-3alpha-ol-17-one | HMDB | | Androsterone | HMDB |
|
|---|
| Chemical Formula | C19H30O2 |
|---|
| Average Molecular Weight | 290.4403 |
|---|
| Monoisotopic Molecular Weight | 290.224580204 |
|---|
| IUPAC Name | (1S,2S,5R,7S,10R,11S,15S)-5-hydroxy-2,15-dimethyltetracyclo[8.7.0.0^{2,7}.0^{11,15}]heptadecan-14-one |
|---|
| Traditional Name | (1S,2S,5R,7S,10R,11S,15S)-5-hydroxy-2,15-dimethyltetracyclo[8.7.0.0^{2,7}.0^{11,15}]heptadecan-14-one |
|---|
| CAS Registry Number | 481-29-8 |
|---|
| SMILES | [H][C@@]12CCC(=O)[C@@]1(C)CC[C@@]1([H])[C@@]2([H])CC[C@@]2([H])C[C@H](O)CC[C@]12C |
|---|
| InChI Identifier | InChI=1S/C19H30O2/c1-18-9-7-13(20)11-12(18)3-4-14-15-5-6-17(21)19(15,2)10-8-16(14)18/h12-16,20H,3-11H2,1-2H3/t12-,13+,14-,15-,16-,18-,19-/m0/s1 |
|---|
| InChI Key | QGXBDMJGAMFCBF-HLUDHZFRSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as androgens and derivatives. These are 3-hydroxylated C19 steroid hormones. They are known to favor the development of masculine characteristics. They also show profound effects on scalp and body hair in humans. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Lipids and lipid-like molecules |
|---|
| Class | Steroids and steroid derivatives |
|---|
| Sub Class | Androstane steroids |
|---|
| Direct Parent | Androgens and derivatives |
|---|
| Alternative Parents | |
|---|
| Substituents | - Androgen-skeleton
- 3-hydroxysteroid
- 3-alpha-hydroxysteroid
- Oxosteroid
- 17-oxosteroid
- Hydroxysteroid
- Cyclic alcohol
- Secondary alcohol
- Ketone
- Organic oxygen compound
- Organooxygen compound
- Hydrocarbon derivative
- Carbonyl group
- Organic oxide
- Alcohol
- Aliphatic homopolycyclic compound
|
|---|
| Molecular Framework | Aliphatic homopolycyclic compounds |
|---|
| External Descriptors | |
|---|
| Ontology |
|---|
| Status | Detected but not Quantified |
|---|
| Origin | |
|---|
| Biofunction | Not Available |
|---|
| Application | Not Available |
|---|
| Cellular locations | - Cell membrane
- Cytoplasm
- Endoplasmic reticulum
- Membrane
|
|---|
| Physical Properties |
|---|
| State | Solid |
|---|
| Experimental Properties | | Property | Value | Reference |
|---|
| Melting Point | Not Available | Not Available | | Boiling Point | Not Available | Not Available | | Water Solubility | Not Available | Not Available | | LogP | Not Available | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | |
|---|
| GC-MS | GC-MS Spectrum - GC-MS (1 TMS) | splash10-056u-6920000000-1d886b76fe1f65860afb | View in MoNA |
|---|
| GC-MS | GC-MS Spectrum - GC-MS (1 MEOX; 1 TMS) | splash10-00al-9820000000-321baddda5ac4f8adaa7 | View in MoNA |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | View in JSpectraViewer |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_1_1) - 70eV, Positive | Not Available | View in JSpectraViewer |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_1_2) - 70eV, Positive | Not Available | View in JSpectraViewer |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TBDMS_1_1) - 70eV, Positive | Not Available | View in JSpectraViewer |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TBDMS_1_2) - 70eV, Positive | Not Available | View in JSpectraViewer |
|---|
| LC-MS/MS | LC-MS/MS Spectrum - 40V, Positive | splash10-0536-7900000000-8cce10a8e140584407c3 | View in MoNA |
|---|
| LC-MS/MS | LC-MS/MS Spectrum - 10V, Negative | splash10-000i-0090000000-fbea13833f57d0820cd8 | View in MoNA |
|---|
| LC-MS/MS | LC-MS/MS Spectrum - 40V, Negative | splash10-052r-6490000000-d9f8bf6f9bc4664ccad2 | View in MoNA |
|---|
| LC-MS/MS | LC-MS/MS Spectrum - 20V, Negative | splash10-000i-0090000000-f56a880ba97983c7e021 | View in MoNA |
|---|
| LC-MS/MS | LC-MS/MS Spectrum - 30V, Positive | splash10-0bta-0930000000-49751ee4f183943f62fa | View in MoNA |
|---|
| LC-MS/MS | LC-MS/MS Spectrum - 10V, Positive | splash10-0006-0090000000-f81b1315d55db1d47ec4 | View in MoNA |
|---|
| LC-MS/MS | LC-MS/MS Spectrum - 35V, Positive | splash10-001c-7190000000-e870cf89a876e5f81d31 | View in MoNA |
|---|
| LC-MS/MS | LC-MS/MS Spectrum - 20V, Positive | splash10-0a4i-0390000000-85c48da18cebca66ba3c | View in MoNA |
|---|
| LC-MS/MS | LC-MS/MS Spectrum - 55V, Positive | splash10-00di-0090000000-4a8b942c855c02555969 | View in MoNA |
|---|
| LC-MS/MS | LC-MS/MS Spectrum - 30V, Positive | splash10-0bta-0930000000-078d7455103eab0cf1ee | View in MoNA |
|---|
| LC-MS/MS | LC-MS/MS Spectrum - 10V, Positive | splash10-05fr-0390000000-905b691e9dad87bc603c | View in MoNA |
|---|
| LC-MS/MS | LC-MS/MS Spectrum - 35V, Positive | splash10-00di-0090000000-514df1ac9844abce75a9 | View in MoNA |
|---|
| LC-MS/MS | LC-MS/MS Spectrum - 20V, Positive | splash10-0a4j-1930000000-b9c49a039799b0a23e04 | View in MoNA |
|---|
| LC-MS/MS | LC-MS/MS Spectrum - 40V, Positive | splash10-08gj-0900000000-9f326ea5c1c454997fd3 | View in MoNA |
|---|
| LC-MS/MS | LC-MS/MS Spectrum - 50V, Positive | splash10-02cv-0900000000-9d78808dd524e4ca6c50 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-00dl-0090000000-42eb8c941695a061666d | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-00dl-0290000000-1a37c17eb6ea7be9211c | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-06u7-2790000000-d6e7cebd4af538181f77 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-000i-0090000000-9e9f9de3b33af0311acd | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-000i-0090000000-5ba62f51f815ded16002 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0596-2190000000-513ad65531fff88b9f69 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-000i-0090000000-c8c452fd99c6bb4f3631 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-000i-0090000000-c8c452fd99c6bb4f3631 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-000i-0090000000-00c0e7de4e528b98df5c | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0006-0090000000-afb176951034ef62419f | View in MoNA |
|---|
|
|---|