| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-09-30 23:30:01 UTC |
|---|
| Update Date | 2020-05-21 16:27:32 UTC |
|---|
| BMDB ID | BMDB0007076 |
|---|
| Secondary Accession Numbers | |
|---|
| Metabolite Identification |
|---|
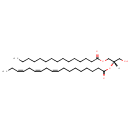
| Common Name | DG(15:0/18:3(9Z,12Z,15Z)/0:0) |
|---|
| Description | DG(15:0/18:3(9Z,12Z,15Z)/0:0)[iso2], also known as dg(15:0/18:3(9z,12z,15z)/0:0)[iso2] or DAG(15:0/18:3), belongs to the class of organic compounds known as lineolic acids and derivatives. These are derivatives of lineolic acid. Lineolic acid is a polyunsaturated omega-6 18 carbon long fatty acid, with two CC double bonds at the 9- and 12-positions. Thus, DG(15:0/18:3(9Z,12Z,15Z)/0:0)[iso2] is considered to be a diradylglycerol lipid molecule. DG(15:0/18:3(9Z,12Z,15Z)/0:0)[iso2] is a very hydrophobic molecule, practically insoluble (in water), and relatively neutral. In cattle, DG(15:0/18:3(9Z,12Z,15Z)/0:0)[iso2] is involved in a couple of metabolic pathways, which include de novo triacylglycerol biosynthesis TG(15:0/18:3(9Z,12Z,15Z)/18:4(6Z,9Z,12Z,15Z)) pathway and de novo triacylglycerol biosynthesis TG(15:0/18:3(9Z,12Z,15Z)/20:2(11Z,14Z)) pathway. |
|---|
| Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| DAG(33:3) | Lipid Annotator, HMDB | | DG(33:3) | Lipid Annotator, HMDB | | DG(15:0/18:3(9Z,12Z,15Z)/0:0) | Lipid Annotator | | Diacylglycerol(15:0/18:3) | Lipid Annotator, HMDB | | Diglyceride | Lipid Annotator, HMDB | | Diacylglycerol | Lipid Annotator, HMDB | | DAG(15:0/18:3) | Lipid Annotator, HMDB | | DG(15:0/18:3) | Lipid Annotator, HMDB | | Diacylglycerol(33:3) | Lipid Annotator, HMDB | | 1-pentadecanoyl-2-(9Z,12Z,15Z-octadeatrienoyl)-sn-glycerol | Lipid Annotator, HMDB | | 1-pentadecanoyl-2-a-linolenoyl-sn-glycerol | Lipid Annotator, HMDB | | 1-Pentadecanoyl-2-alpha-linolenoyl-sn-glycerol | HMDB | | DAG(15:0/18:3N3) | HMDB | | DAG(15:0/18:3W3) | HMDB | | DG(15:0/18:3N3) | HMDB | | DG(15:0/18:3W3) | HMDB | | Diacylglycerol(15:0/18:3n3) | HMDB | | Diacylglycerol(15:0/18:3W3) | HMDB |
|
|---|
| Chemical Formula | C36H64O5 |
|---|
| Average Molecular Weight | 576.8904 |
|---|
| Monoisotopic Molecular Weight | 576.475375158 |
|---|
| IUPAC Name | (2S)-1-hydroxy-3-(pentadecanoyloxy)propan-2-yl (9Z,12Z,15Z)-octadeca-9,12,15-trienoate |
|---|
| Traditional Name | diacylglycerol |
|---|
| CAS Registry Number | Not Available |
|---|
| SMILES | [H][C@](CO)(COC(=O)CCCCCCCCCCCCCC)OC(=O)CCCCCCC\C=C/C\C=C/C\C=C/CC |
|---|
| InChI Identifier | InChI=1S/C36H64O5/c1-3-5-7-9-11-13-15-17-18-19-21-23-25-27-29-31-36(39)41-34(32-37)33-40-35(38)30-28-26-24-22-20-16-14-12-10-8-6-4-2/h5,7,11,13,17-18,34,37H,3-4,6,8-10,12,14-16,19-33H2,1-2H3/b7-5-,13-11-,18-17-/t34-/m0/s1 |
|---|
| InChI Key | UARHXHPRJDJCKE-QECOYPFHSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as lineolic acids and derivatives. These are derivatives of lineolic acid. Lineolic acid is a polyunsaturated omega-6 18 carbon long fatty acid, with two CC double bonds at the 9- and 12-positions. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Lipids and lipid-like molecules |
|---|
| Class | Fatty Acyls |
|---|
| Sub Class | Lineolic acids and derivatives |
|---|
| Direct Parent | Lineolic acids and derivatives |
|---|
| Alternative Parents | |
|---|
| Substituents | - Octadecanoid
- 1,2-acyl-sn-glycerol
- Diacylglycerol
- Diradylglycerol
- Fatty acid ester
- Glycerolipid
- Dicarboxylic acid or derivatives
- Carboxylic acid ester
- Carboxylic acid derivative
- Organooxygen compound
- Hydrocarbon derivative
- Organic oxygen compound
- Organic oxide
- Carbonyl group
- Primary alcohol
- Alcohol
- Aliphatic acyclic compound
|
|---|
| Molecular Framework | Aliphatic acyclic compounds |
|---|
| External Descriptors | |
|---|
| Ontology |
|---|
| Status | Expected but not Quantified |
|---|
| Origin | |
|---|
| Biofunction | Not Available |
|---|
| Application | Not Available |
|---|
| Cellular locations | - Cell membrane
- Cytoplasm
- Membrane
|
|---|
| Physical Properties |
|---|
| State | Solid |
|---|
| Experimental Properties | | Property | Value | Reference |
|---|
| Melting Point | Not Available | Not Available | | Boiling Point | Not Available | Not Available | | Water Solubility | Not Available | Not Available | | LogP | Not Available | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (1 TMS) - 70eV, Positive | splash10-000t-6962204000-693f70bf6366a395371b | View in MoNA |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS ("DG(15:0/18:3(9Z,12Z,15Z)/0:0),1TMS,#1" TMS) - 70eV, Positive | Not Available | View in JSpectraViewer |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TBDMS_1_1) - 70eV, Positive | Not Available | View in JSpectraViewer |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0006-0000090000-1cbb27a90e650523547b | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-052t-0099090000-eca62e280c5595793e4d | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-000n-0099090000-a6866049c39b7067d929 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0002-0000090000-70867ade9b27f237daa1 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0002-0000090000-70867ade9b27f237daa1 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0ai0-0009000000-c275f36e2b4e95468f57 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0006-0000090000-1dffb1eae7d06d9e56e5 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-052t-0088090000-99b936e00e4e72c0c7c6 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-000n-0088090000-e6633f8c1ee15aa1e4fd | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-004i-1166390000-f8d4e3ef3a70c7352587 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-08g0-4292010000-5f27b8fd494dee5b3c67 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-08i9-6795000000-6e42aef54995b4594d3e | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-004i-2094080000-c5cc0cdc1ab3e6a35c98 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-006x-2090000000-d11e45bc577845051ee0 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-00bc-2190000000-eec2760e5a2a689c5e47 | View in MoNA |
|---|
|
|---|
| Pathways | |
|---|