| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-09-30 22:45:21 UTC |
|---|
| Update Date | 2020-05-21 16:26:56 UTC |
|---|
| BMDB ID | BMDB0001396 |
|---|
| Secondary Accession Numbers | |
|---|
| Metabolite Identification |
|---|
| Common Name | 5-Methyltetrahydrofolic acid |
|---|
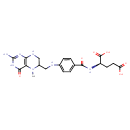
| Description | 5-Methyltetrahydrofolic acid, also known as 5-methyltetrahydrofolic acid or 5-methyltetrahydrofolic acid, belongs to the class of organic compounds known as tetrahydrofolic acids. These are heterocyclic compounds based on the 5,6,7,8-tetrahydropteroic acid skeleton conjugated with at least one L-glutamic acid unit. 5-Methyltetrahydrofolic acid is possibly soluble (in water) and a strong basic compound (based on its pKa). 5-Methyltetrahydrofolic acid exists in all eukaryotes, ranging from yeast to humans. 5-Methyltetrahydrofolic acid participates in a number of enzymatic reactions, within cattle. In particular, Homocysteine and 5-methyltetrahydrofolic acid can be converted into L-methionine and tetrahydrofolic acid through its interaction with the enzyme methionine synthase. In addition, 5-Methyltetrahydrofolic acid can be biosynthesized from 5,10-methylene-THF; which is catalyzed by the enzyme methylenetetrahydrofolate reductase. In cattle, 5-methyltetrahydrofolic acid is involved in a couple of metabolic pathways, which include the betaine metabolism pathway and the folate metabolism pathway. |
|---|
| Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 5-Methyltetrahydrofolate | Generator | | 5-Methyl tetrahydrofolate | HMDB | | 5-Methyl-5,6,7,8-tetrahydrofolate | HMDB | | 5-Methyl-tetrahydrofolate | HMDB | | 5-Methyltetrahydropteroylglutamate | HMDB | | Methyl folate | HMDB | | Methyl-tetrahydrofolate | HMDB | | N( 5)-Methyltetrahydrofolate | HMDB | | N-(4-(((2-Amino-1,4,5,6,7,8-hexahydro-5-methyl-4-oxo-6-pteridinyl)methyl)amino)benzoyl)-L-glutamate | HMDB | | N-(4-(((2-Amino-1,4,5,6,7,8-hexahydro-5-methyl-4-oxo-6-pteridinyl)methyl)amino)benzoyl)-L-glutamic acid | HMDB | | N-(5-Methyl-5,6,7,8-tetrahydropteroyl)-L-glutamate | HMDB | | N-(5-Methyl-5,6,7,8-tetrahydropteroyl)-L-glutamic acid | HMDB | | N5-Methyl-tetrahydrofolate | HMDB | | N5-Methyl-tetrahydrofolic acid | HMDB | | N5-Methyltetrahydrofolate | HMDB | | N5-Methyltetrahydropteroyl mono-L-glutamate | HMDB | | [(6S)-5-Methyl-5,6,7,8-tetrahydropteroyl]glutamate | HMDB | | 5-Methyltetrahydrofolate, (DL-glu)-isomer | HMDB | | 5-Methyltetrahydrofolate, (L-glu)-(R)-isomer | HMDB | | 5-Methyltetrahydrofolate, (L-glu)-(S)-isomer | HMDB | | 5-Methyltetrahydrofolate, calcium salt (1:1), (L-glu)-isomer | HMDB | | 5-Methyltetrahydrofolate, methyl-(14)C-labeled, (DL-glu)-isomer | HMDB | | 5-Methyltetrahydrofolate, methyl-(14)C-labeled, (L-glu)-isomer | HMDB | | CH3-FH4 | HMDB | | L-Methyl folate | HMDB | | N(5)-Methyltetrahydrofolic acid | HMDB | | Prefolic a | HMDB | | Mefolinate | HMDB | | Levomefolic acid | HMDB | | L-Methylfolate | HMDB | | Deplin | HMDB |
|
|---|
| Chemical Formula | C20H25N7O6 |
|---|
| Average Molecular Weight | 459.4558 |
|---|
| Monoisotopic Molecular Weight | 459.186631567 |
|---|
| IUPAC Name | (2R)-2-[(4-{[(2-amino-5-methyl-4-oxo-3,4,5,6,7,8-hexahydropteridin-6-yl)methyl]amino}phenyl)formamido]pentanedioic acid |
|---|
| Traditional Name | (2R)-2-[(4-{[(2-amino-5-methyl-4-oxo-3,6,7,8-tetrahydropteridin-6-yl)methyl]amino}phenyl)formamido]pentanedioic acid |
|---|
| CAS Registry Number | 134-35-0 |
|---|
| SMILES | CN1C(CNC2=CC=C(C=C2)C(=O)N[C@H](CCC(O)=O)C(O)=O)CNC2=C1C(=O)NC(N)=N2 |
|---|
| InChI Identifier | InChI=1S/C20H25N7O6/c1-27-12(9-23-16-15(27)18(31)26-20(21)25-16)8-22-11-4-2-10(3-5-11)17(30)24-13(19(32)33)6-7-14(28)29/h2-5,12-13,22H,6-9H2,1H3,(H,24,30)(H,28,29)(H,32,33)(H4,21,23,25,26,31)/t12?,13-/m1/s1 |
|---|
| InChI Key | ZNOVTXRBGFNYRX-ZGTCLIOFSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as tetrahydrofolic acids. These are heterocyclic compounds based on the 5,6,7,8-tetrahydropteroic acid skeleton conjugated with at least one L-glutamic acid unit. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organoheterocyclic compounds |
|---|
| Class | Pteridines and derivatives |
|---|
| Sub Class | Pterins and derivatives |
|---|
| Direct Parent | Tetrahydrofolic acids |
|---|
| Alternative Parents | |
|---|
| Substituents | - Tetrahydrofolic acid
- Glutamic acid or derivatives
- Hippuric acid or derivatives
- Hippuric acid
- N-acyl-alpha-amino acid
- N-acyl-alpha amino acid or derivatives
- Alpha-amino acid or derivatives
- Aminobenzamide
- Aminobenzoic acid or derivatives
- Benzoic acid or derivatives
- Benzamide
- Benzoyl
- Phenylalkylamine
- Tertiary aliphatic/aromatic amine
- Aniline or substituted anilines
- Dialkylarylamine
- Secondary aliphatic/aromatic amine
- Pyrimidone
- Aminopyrimidine
- Dicarboxylic acid or derivatives
- Benzenoid
- Pyrimidine
- Monocyclic benzene moiety
- Vinylogous amide
- Heteroaromatic compound
- Secondary carboxylic acid amide
- Tertiary amine
- Amino acid or derivatives
- Amino acid
- Carboxamide group
- Secondary amine
- Carboxylic acid
- Carboxylic acid derivative
- Azacycle
- Organic nitrogen compound
- Carbonyl group
- Organonitrogen compound
- Organooxygen compound
- Primary amine
- Hydrocarbon derivative
- Organic oxide
- Organopnictogen compound
- Organic oxygen compound
- Amine
- Aromatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aromatic heteropolycyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Ontology |
|---|
| Status | Detected but not Quantified |
|---|
| Origin | |
|---|
| Biofunction | Not Available |
|---|
| Application | Not Available |
|---|
| Cellular locations | Not Available |
|---|
| Physical Properties |
|---|
| State | Solid |
|---|
| Experimental Properties | | Property | Value | Reference |
|---|
| Melting Point | Not Available | Not Available | | Boiling Point | Not Available | Not Available | | Water Solubility | Not Available | Not Available | | LogP | Not Available | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-03di-2645900000-0472550b7df0d95d4718 | View in MoNA |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (2 TMS) - 70eV, Positive | splash10-059i-4532290000-8e3b1526c05a0a70d971 | View in MoNA |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | View in JSpectraViewer |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-03dl-0313900000-d4f9d7f9d8c65a2e0428 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-01ox-0924300000-2f133db2f48bd5b14a8d | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-000f-0920000000-298e8f3cd55b0fe42bad | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0a4i-0001900000-b1b1292a418283f61ea9 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0900-1256900000-32c1cf48970dead32f60 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0006-9641100000-b5468bf27455b7e5ee39 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0a4i-0001900000-ba41f53e73f5102d5c04 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0a4u-3419800000-385454b6189707f5e684 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0zg0-3931100000-ccab9c792ff1e540a4e8 | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-03di-0108900000-f5ef3a5a71a48db8560b | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-03di-0209100000-ac8dd1f905331a1f84bb | View in MoNA |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-000x-0912000000-e973388138222d017517 | View in MoNA |
|---|
| 1D NMR | 13C NMR Spectrum (1D, 100 MHz, D2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 1H NMR Spectrum (1D, 100 MHz, D2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 13C NMR Spectrum (1D, 1000 MHz, D2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 1H NMR Spectrum (1D, 1000 MHz, D2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 13C NMR Spectrum (1D, 200 MHz, D2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 1H NMR Spectrum (1D, 200 MHz, D2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 13C NMR Spectrum (1D, 300 MHz, D2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 1H NMR Spectrum (1D, 300 MHz, D2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 13C NMR Spectrum (1D, 400 MHz, D2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 1H NMR Spectrum (1D, 400 MHz, D2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 13C NMR Spectrum (1D, 500 MHz, D2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 1H NMR Spectrum (1D, 500 MHz, D2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 13C NMR Spectrum (1D, 600 MHz, D2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 1H NMR Spectrum (1D, 600 MHz, D2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 13C NMR Spectrum (1D, 700 MHz, D2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 1H NMR Spectrum (1D, 700 MHz, D2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 13C NMR Spectrum (1D, 800 MHz, D2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 1H NMR Spectrum (1D, 800 MHz, D2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 13C NMR Spectrum (1D, 900 MHz, D2O, predicted) | Not Available | View in JSpectraViewer |
|---|
| 1D NMR | 1H NMR Spectrum (1D, 900 MHz, D2O, predicted) | Not Available | View in JSpectraViewer |
|---|
|
|---|